3 lb of sodium sulfite and 3 lb of caustic soda should be added per 1000 gal of water contained in the boiler minimum 400 ppm P-alkalinity as CaCO3 and 200 ppm sulfite as SO3. Which one of the following best describes the difference between molar mass and formula weight.

Difference Between Sulfur Sulfate And Sulfite Compare The Difference Between Similar Terms
Difference Between Sulfate And Sulfite Definition Properties Examples

Answered 12 What Is The Difference Between Bartleby
Sulfuric acid is by far the most important industrial chemical.
Difference between sulfate and sulfite. Sulfate has a positive charge while sulfite has a negative charge. Hazardous waste and decaying organic material can seep through the soil and cause sulfates to contaminate groundwater. Sulfite has one less sulfur than sulfate.
In its anhydrous form image provided above sodium sulfite is a white solid. Visit BYJUS to learn about the Physical and Chemical Properties of Sodium Thiosulfate along with its Uses. The coordination number of an ion also called its ligancy in a crystal lattice structure is the number of immediate neighbors it has possessing the opposite charge.
Acceleration Formula Force Formula Frequency Formula Velocity Formula Wavelength Formula Angular Velocity Formula Displacement Formula Density Formula Kinematic Equations Formula Tangential Velocity Formula Kinetic Energy Formula Angular Speed Formula Buoyancy Formula Efficiency Formula Static Friction Formula Potential Energy. How many atoms make up the oxalate ion and what is its charge. Sodium Thiosulfate Na2S203 - Sodium thiosulfate is a colorless transparent monoclinic crystal widely used by utilities for dechlorination.
Na 2 SO 37H 2 O is slowly oxidized by atmospheric oxygen giving rise to the corresponding sulfate. S 3 x O -2 S 3 x -2 -2 S -6 -2 S 4 Answer. Note that the -ite polyatomic ion has one less oxygen atom in its formula than the -ate ion but with the same ionic charge.
Both are wine making ingredients and add sulfites to wines. B A formula weight is the mass of one molecule or formula unit while a molar mass is the mass of one mole of molecules or formula units. Water is then added as needed to keep the concentration of this solution between 96 and 98 H 2 SO 4 by weight.
Difference Between Sack and Bag Difference Between Sulfur Sulfate and Sulfite Difference Between List and Tuple Difference Between Annual and Perennial Plants Difference Between Chapter 7. A Formula weights are measured in grams per mole while molar masses are measured in atomic mass units. Structure of Sodium Sulfite- Na 2 SO 3 In a sodium sulfite molecule there.
Another important reaction of sulfur dioxide is with the base calcium oxide to form calcium sulfite also known as calcium sulfateIV. Sulfur in the sulfite ion has an oxidation. It is abundant multivalent and nonmetallicUnder normal conditions sulfur atoms form cyclic octatomic molecules with a chemical formula S 8Elemental sulfur is a bright yellow crystalline solid at room temperature.
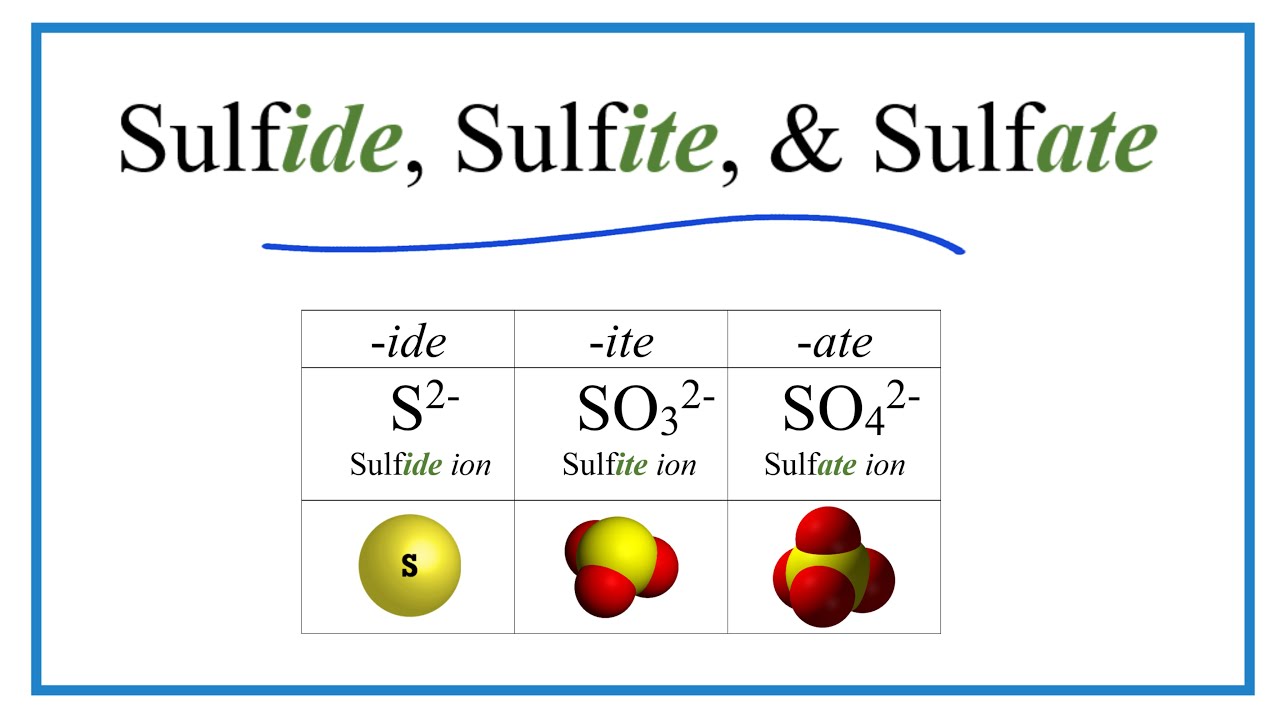
Sulphur is a chemical element with the symbol S and atomic number 16. For example SO 3 2 is called sulfite and SO 4 2 is called sulfate. Some of them such as nitrate and nitrite and sulfate and sulfite have very similar formulas and names so care must be taken to get the formulas and names correct.
Zinc sulfate is used to treat and preserve wood while sodium sulfate is used during the manufacture of paper and glass. What is the difference between sulfate and sulfite. 5 lb of a 35 solution of hydrazine and 01 lb of ammonia or 2-3 lb of a 40 solution of neutralizing amine can be added per 1000 gal minimum 200 ppm hydrazine and 100 pH.
The primary difference between anhydrous Na 2 SO 3 and its heptahydrate is the relative stability of the anhydrous form towards oxidation. Sulfite is a potentially toxic molecule that might enter the body via ingestion inhalation or injection. Sodium sulfite Na 2 SO 3 210 g Basic fuchsin 105 g Agar optional 150 g Reagent-grade water 1 L 1 Agar preparationRehydrate product in 1 L water containing 20 mL 95 ethanol.
The latter is usually preferred for the following three reasons. There is no difference they are the same thing. Heat to near boiling to dissolve agar then promptly remove from heat and cool to between 45 and 50C.
The eletronegativity difference between two atoms. The acute and subacute toxicities of several CrIII and CrVI compounds chromium3 chloride chromium3 nitrate chromium3 sulfate chromium trioxide potassium dichromate were determined in NZC and CxO mice injected ipThe distal median lethal doses 10 days after treatment averaged 179 or - 18 X 10-6 g chromiumg body wt regardless of the oxidation state of the Cr. Sulfur in nontechnical British English.
It has even been argued that there is a direct relationship between the amount of sulfuric acid a. Just like the previous example oxygen has an oxidation state of -2 and the total oxidation of the ion is -2. In those cases where more than two oxy anions constitute the series hypo- less than and per- more than are used as prefixes to name the members of the series with the smallest and the largest number of oxygen atoms respectively.
Forty normal and sulfite oxidase-deficient male albino rats. The only difference is the one less oxygen. Note that the -ite polyatomic ion has one less oxygen atom in its formula than the -ate ion but with the same ionic charge.
Not bringing any dietary intake of sodium which will raise the blood pressure. Some of them such as nitrate and nitrite and sulfate and sulfite have very similar formulas and names so care must be taken to get the formulas and names correct. Sulfate occasionally becomes a water contaminant under the right conditions.
The greater the difference the greater the attraction between the two atoms. Sulfate was detected as a photoredox co-product 14 by precipitation of barium sulfate on the addition of barium chloride under conditions in which barium sulfite is soluble 17. What is Sulfate Definition Properties Examples 2.
The main difference between them is sodium metabisulfite will add sodium ions in the wine while potassium metabisulfite will leave potassium ions. 637 Write the correct formula for the following ionic compounds. Sulfur is the tenth most common element by mass in the universe.
Non-antibiotic eye drops like Genoptic gentamicin sulfate Isopto Atropine atropine sulfate Maxitrol neomycin dexamethasone polymyxin B and Polytrim polymyxin B trimethoprim Oral sulfonylureas-class drugs like Amaryl glimepiride Glynase glyburide and Glucotrol glipizide used to treat type 2 diabetes. The main difference between sulfate and sulfite is that sulfate is composed of four oxygen atoms bonded to a sulfur atom whereas sulfite is composed of three oxygen atoms bonded to a sulfur atom. What three hydrogen-containing polyatomic anions are essential components of living.
This is possible because the electronegativity difference between aluminum and oxygen is small unlike the difference between sodium and oxygen for example. The anion to cation ratio must be reflected in this coordination number which can influence the color of the compound. What is the oxidation state of the sulfur atom in the sulfite ion - SO 3 2-.
Circle the letter of a polyatomic ion that is a cation. For cellular detoxification mammalians rely on sulfite oxidase to convert sulfite to sulfateThe purpose of this research was to determine the effect of sulfite on zinc iron and copper levels in rat liver and kidney tissues. Look at Table 93.
Sulfate has one more oxygen than sulfite. What is the difference between the sulfite and sulfate anions.

Reactions Of Oxidation Of Sulfite Radical To Peroxymonosulfate And Download Scientific Diagram

In Situ Drifts Spectra Of O 3 Oxidation Of Sulfite Mixed Into Caco 3 Download Scientific Diagram

Production Of Dimethyl Sulfoxide From Lignin Team Bravo Is Jake Biberstein Stan Das Jeff Umbach Russ Boyer Krista Sutton Mike Czepiak Project Lead Ppt Download

Difference Between Kraft And Sulfite Pulping Compare The Difference Between Similar Terms
How Does Sulfate And Sulfite Differ Quora
Sulfide Sulfite Sulfate Ions Difference And Formulas Youtube
Sulfate And Sulfite I 524a

Answered Difference Between Sulphate Sulphite Sulphide Brainly In

